Classification of Carbon and Graphite
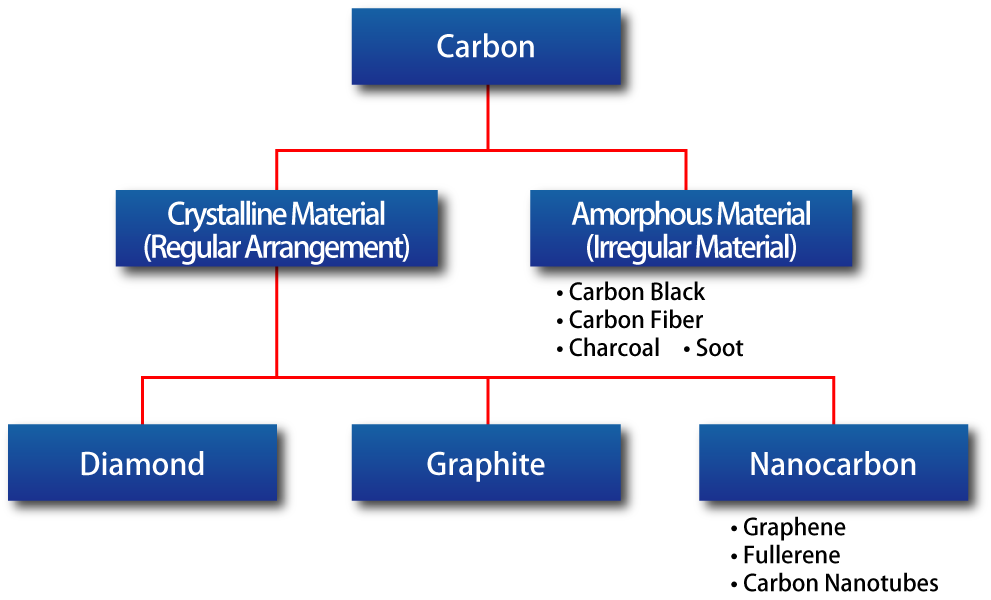
Graphite is a form of carbon with a well-developed crystalline structure.
Types of Graphite
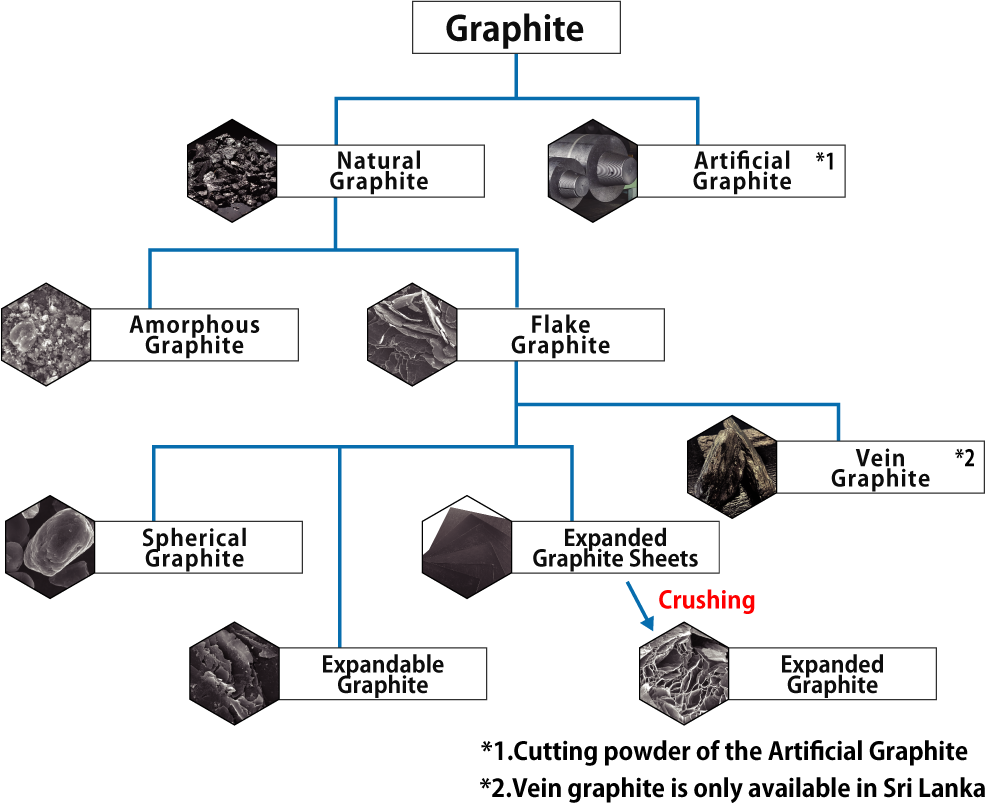
There are two types of graphite: natural and artificial. Natural graphite is roughly categorized into two types depending on the crystalline state: flake graphite and amorphous graphite. Flake graphite is silvery-black and appears as thin flakes, while amorphous graphite is black and looks like soil.
Artificial graphite is made from coke and pitch by artificially developing graphite crystals under extremely high temperatures and is mainly used as electrodes for steel manufacturing.
Flake Graphite Manufacturing Process
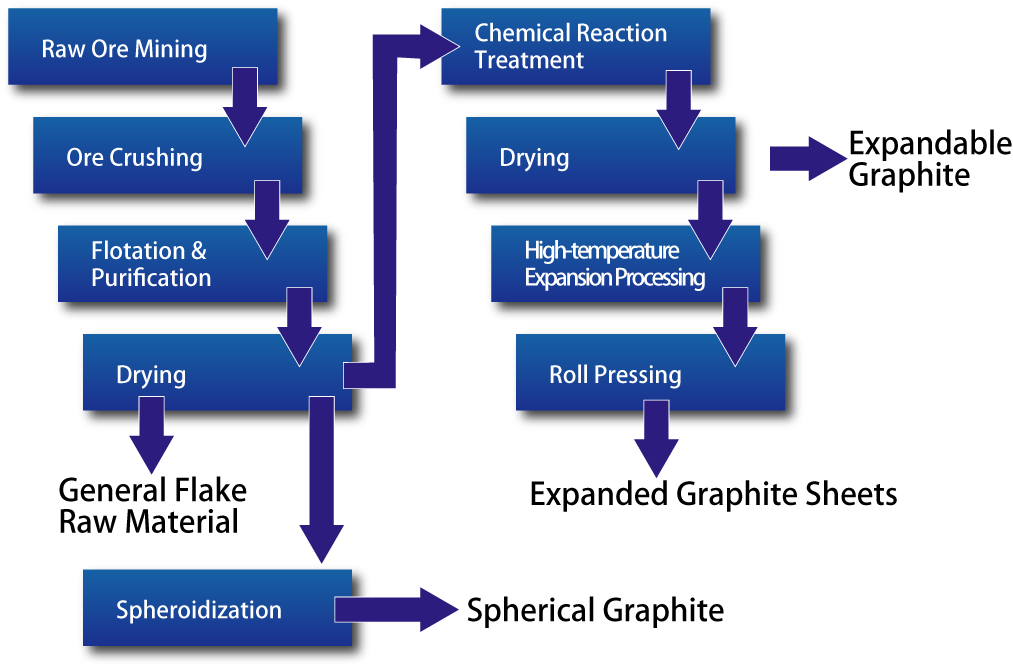
Among the different types of graphite, flake graphite is used to manufacture various products.
At Nishimura Graphite, more than half of our product lineup is derived from flake graphite.
Global Natural Graphite Mines
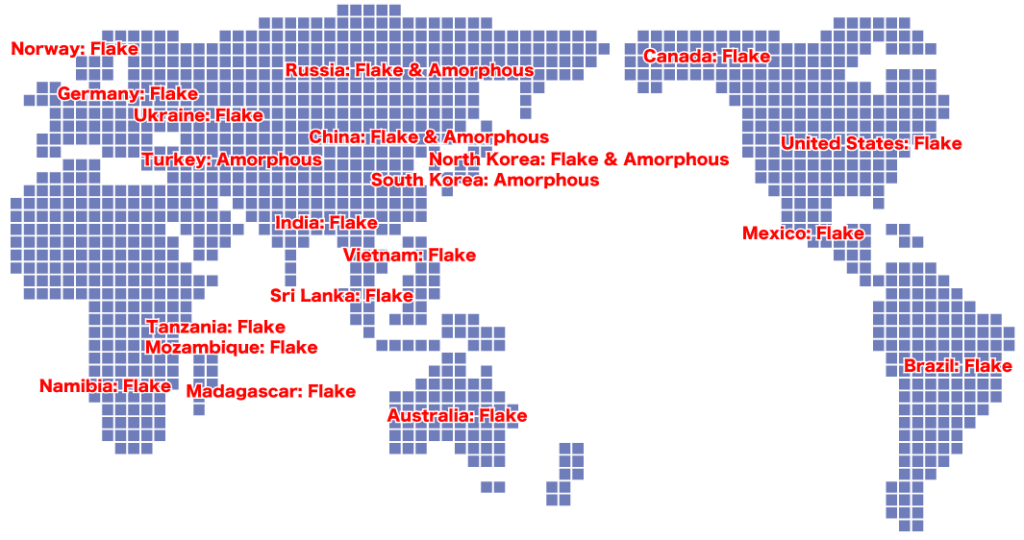
As shown on this map, there are many natural graphite mines around the world.
While most of the graphite imported into Japan is produced in China, we also secure raw material supply routes from other countries to mitigate geopolitical risks.
Five Key Properties of Graphite

Graphite possesses these five key properties.
Together, these properties further enhance the value of graphite.
- Heat Resistance
Withstands up to 3550°C in a reducing atmosphere (where oxygen is not present).
In an oxidizing atmosphere (where oxygen is present), it begins turning to ash at around 500°C.
Sublimates and turns into a gas at temperatures above 3550°C (liquid graphite does not exist).
Does not soften or melt. It gains strength at higher temperatures.
Used in high-temperature environments such as furnaces.
- Lubrication
Lubricates by allowing its crystal layers to slide over each other.
Exhibits self-lubricating properties, making it usable without oil.
Functions as a solid lubricant that is effective even at high temperatures.
Used as a friction modifier in brake pads, etc.
- Electrical Conductivity
Added as an antistatic filler in various materials.
According to academic literature, it has a theoretical electric conductivity of 10-4Ωcm.
Also used as electromagnetic shielding material.
Used in conductive coatings, etc.
- Heat Conductivity
Lighter and more resistant to oxidation compared to typical metals used for heat-dissipating applications
Also used as an alternative to metals in areas where weight reduction is required.
Used as an additive in heat-dissipating resins, etc.
- Chemical Resistance
Prevents deterioration of parts exposed to various chemicals.
The various types of graphite are chemically stable, making them less reactive to strong acids and alkalis.
Used in protective coatings, etc.
